PD-L1 testing is an important biomarker assessment used to guide the use of immunotherapy in several cancers, particularly lung cancer and other solid tumors. By measuring PD-L1 expression on tumor cells or immune cells, clinicians can estimate the likelihood that a patient will respond to immune checkpoint inhibitors. While PD-L1 levels do not perfectly predict response, the test helps stratify patients and refine treatment strategy within the broader framework of precision oncology.
In modern oncology practice, immunotherapy is not initiated blindly. Biomarker testing—including PD-L1 expression—is part of a structured molecular evaluation designed to match the right therapy to the right patient.
Perspective from clinical oncology practice: Treatment selection increasingly depends on molecular and immunologic markers rather than tumor location alone.
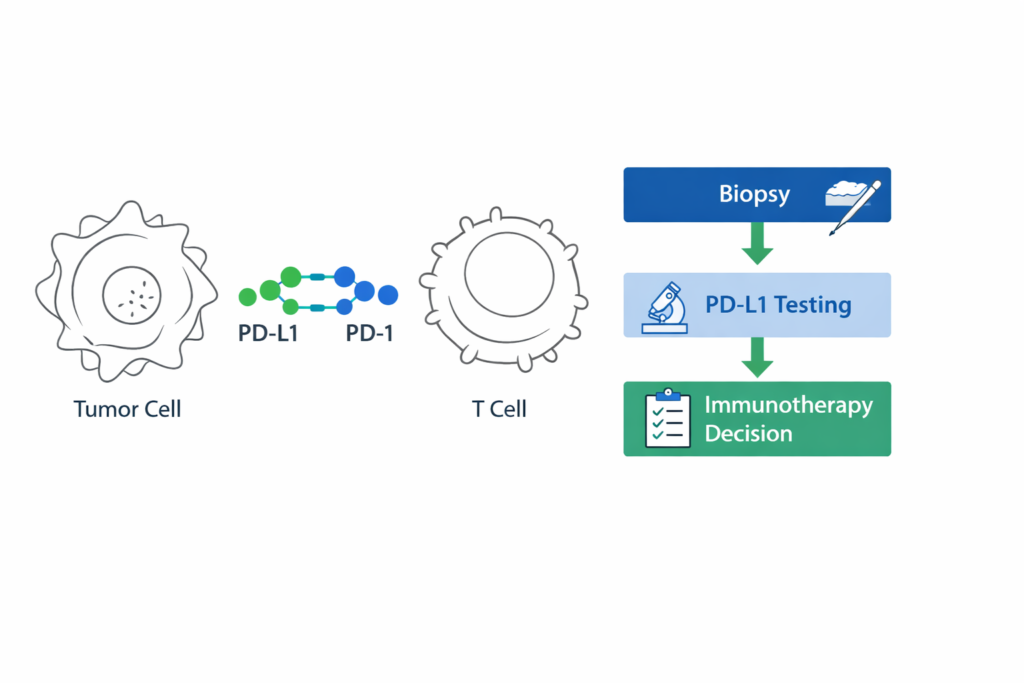
What Is PD-L1 and Why Is It Important in Cancer Treatment?
PD-L1 (Programmed Death Ligand-1) is a protein expressed on the surface of some tumor cells and immune cells. It interacts with the PD-1 receptor on T-cells, a mechanism that tumors can exploit to suppress the immune response.
Immune checkpoint inhibitors work by blocking this interaction, allowing the immune system to recognize and attack cancer cells more effectively.
- PD-L1 expression reflects immune evasion mechanisms within the tumor microenvironment.
- Checkpoint inhibitors target the PD-1 / PD-L1 pathway.
- Higher PD-L1 levels may increase probability of response to immunotherapy.
Which Cancers Commonly Require PD-L1 Testing?
PD-L1 testing is routinely considered in multiple tumor types where immunotherapy plays a major therapeutic role.
- Non-small cell lung cancer (NSCLC)
- Triple-negative breast cancer
- Head and neck cancers
- Gastric and gastro-esophageal cancers
- Urothelial cancers
However, interpretation of PD-L1 expression varies depending on tumor type, testing platform, and scoring system.
How Is PD-L1 Testing Performed?
PD-L1 testing is performed on tumor tissue obtained during biopsy or surgery. The most common method used is immunohistochemistry (IHC), where antibodies detect PD-L1 expression on tumor or immune cells.
- Tissue sample obtained through biopsy
- Immunohistochemistry staining for PD-L1
- Expression measured using tumor proportion score (TPS) or combined positive score (CPS)
Results are typically reported as a percentage indicating the proportion of cells expressing PD-L1.
How PD-L1 Results Influence Treatment Decisions
PD-L1 testing does not act as an isolated decision point. Instead, it is interpreted alongside tumor genetics, disease stage, and patient characteristics.
- Molecular mutation status
- Tumor histology
- Disease stage
- Overall health and treatment goals
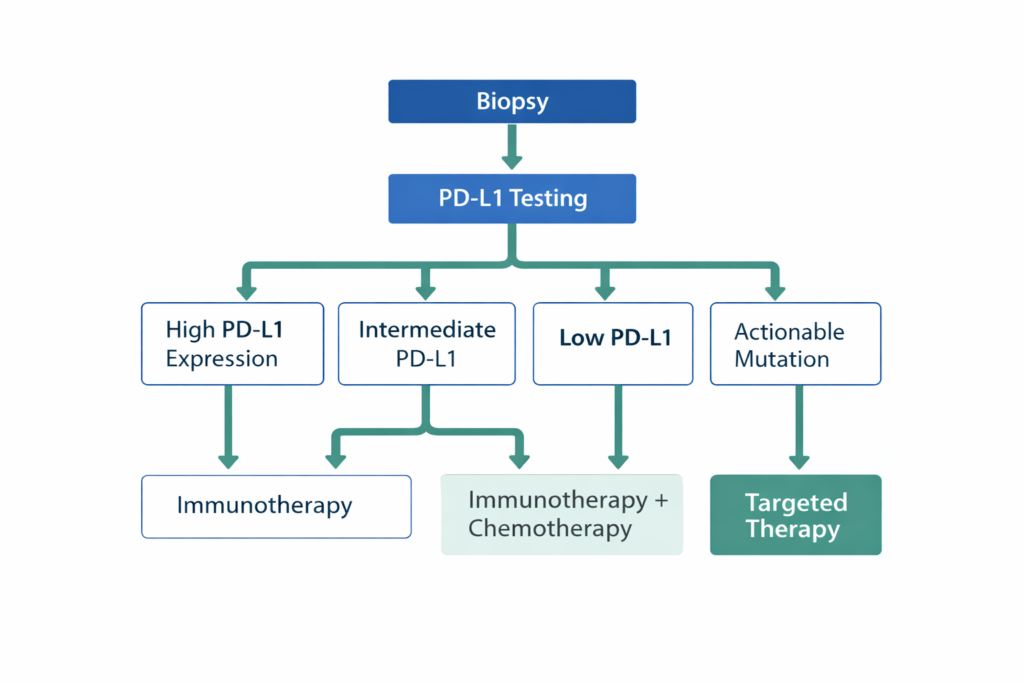
Clinical Decision Logic
- If PD-L1 expression is high → Immunotherapy may be used as first-line treatment in certain cancers.
- If PD-L1 expression is intermediate → Combination therapy (immunotherapy + chemotherapy) may be considered.
- If PD-L1 expression is low or negative → Standard chemotherapy or targeted therapy may be prioritized.
- If actionable genetic mutation is detected → Targeted therapy may take precedence over immunotherapy.
Why Structured Review of Biomarker Reports Matters
PD-L1 interpretation can vary depending on testing methodology and clinical context. In complex cases, a structured second opinion may help clarify how biomarker results should influence treatment selection.
Reviewing pathology slides, molecular testing results, and PD-L1 scoring together ensures that treatment decisions are aligned with precision oncology principles.
Frequently Asked Questions About PD-L1 Testing
Does a high PD-L1 level guarantee that immunotherapy will work?
No. While higher PD-L1 expression increases the likelihood of response, some patients with low PD-L1 levels can still benefit from immunotherapy.
Can PD-L1 levels change over time?
Yes. Tumor biology evolves, and PD-L1 expression may change after treatment or disease progression.
Is PD-L1 testing required for all cancers?
No. Its clinical relevance varies by tumor type and available treatment options.
Is PD-L1 testing the only biomarker needed before immunotherapy?
No. Comprehensive molecular testing and clinical evaluation are also important.
Does a negative PD-L1 test rule out immunotherapy?
Not necessarily. Some treatment regimens include immunotherapy even in PD-L1 negative tumors depending on clinical context.
Related Articles
When Immunotherapy Works Even With Low PD-L1 Expression
Individualizing Cancer Treatment in the Elderly and Young Adults
How Molecular Profiling Influences Treatment Decisions in Lung Cancer
Closing Perspective
PD-L1 testing represents one component of a larger shift toward biomarker-guided cancer treatment. As precision oncology continues to evolve, integrating immunologic markers with molecular profiling allows clinicians to tailor therapy with greater accuracy.
The future of cancer care increasingly lies in combining genomic, immunologic, and clinical data to design individualized treatment strategies.
Educational Disclaimer
This article is intended for educational purposes and should not replace individualized medical advice. Treatment decisions should always be made in consultation with a qualified oncology specialist.
Pingback: Immunotherapy in Low Pd-L1 expression
Pingback: Molecular Subtypes in Cancer:Biology Matters More Than Stage