Genetic testing for cancer risk should be considered when personal or family history suggests a hereditary cancer syndrome. This includes early-onset cancers, multiple affected relatives, rare tumor types, or known pathogenic mutations in the family. Testing helps identify inherited mutations that may influence screening strategies, preventive decisions, and treatment planning.
Not all cancers are hereditary. However, identifying high-risk individuals allows structured surveillance and risk-reduction strategies before cancer develops.
In clinical oncology practice, structured hereditary risk assessment is an integral part of responsible cancer care and precision prevention.
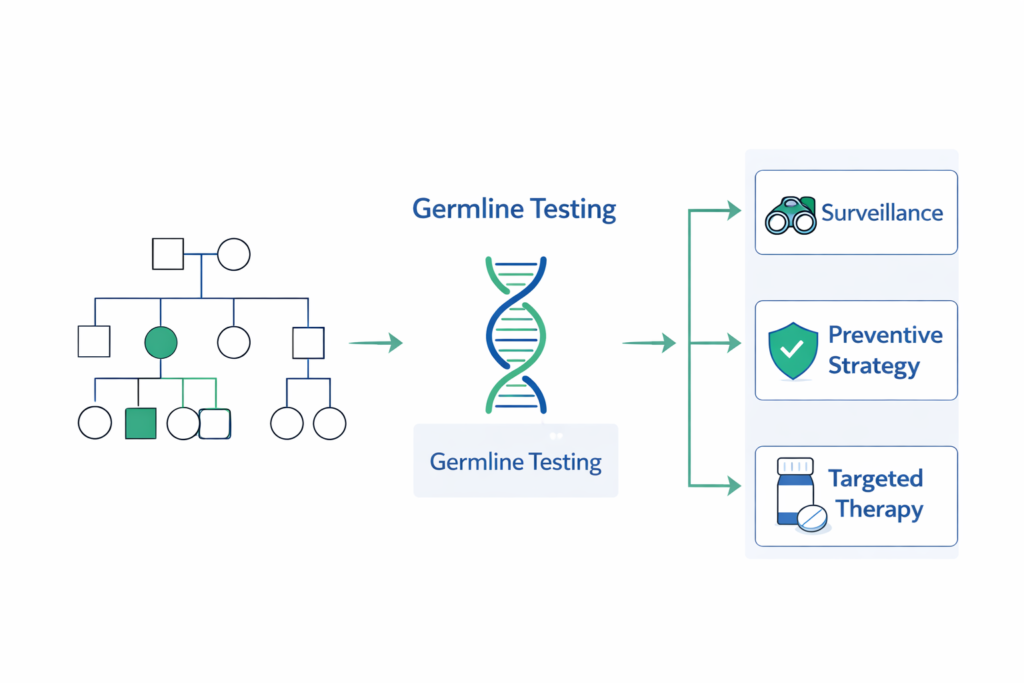
What Is Hereditary Cancer Risk?
Most cancers occur sporadically. However, approximately 5–10% are linked to inherited genetic mutations passed through families.
- BRCA1 / BRCA2 mutations (breast and ovarian cancer)
- Lynch syndrome (colorectal and endometrial cancer)
- TP53 mutations (Li-Fraumeni syndrome)
- Familial adenomatous polyposis (APC mutations)
These inherited variants significantly increase lifetime cancer risk compared to the general population.
Which Clinical Situations Warrant Genetic Testing?
Testing is recommended when clinical red flags suggest hereditary predisposition.
- Cancer diagnosed at an unusually young age
- Multiple primary cancers in one individual
- Two or more close relatives with related cancers
- Male breast cancer
- Rare tumor types associated with syndromes
Genetic counseling should precede and follow testing to ensure accurate interpretation.
Structured If → Then Genetic Testing Logic
- If strong family history present → Then refer for genetic counseling.
- If early-onset cancer (<50 years in many tumor types) → Then evaluate for germline testing.
- If known pathogenic mutation in family → Then offer targeted testing to relatives.
- If tumor shows mismatch repair deficiency → Then assess for Lynch syndrome.
This structured framework avoids over-testing while ensuring high-risk individuals are appropriately evaluated.
Decision Tree for Genetic Testing in Oncology
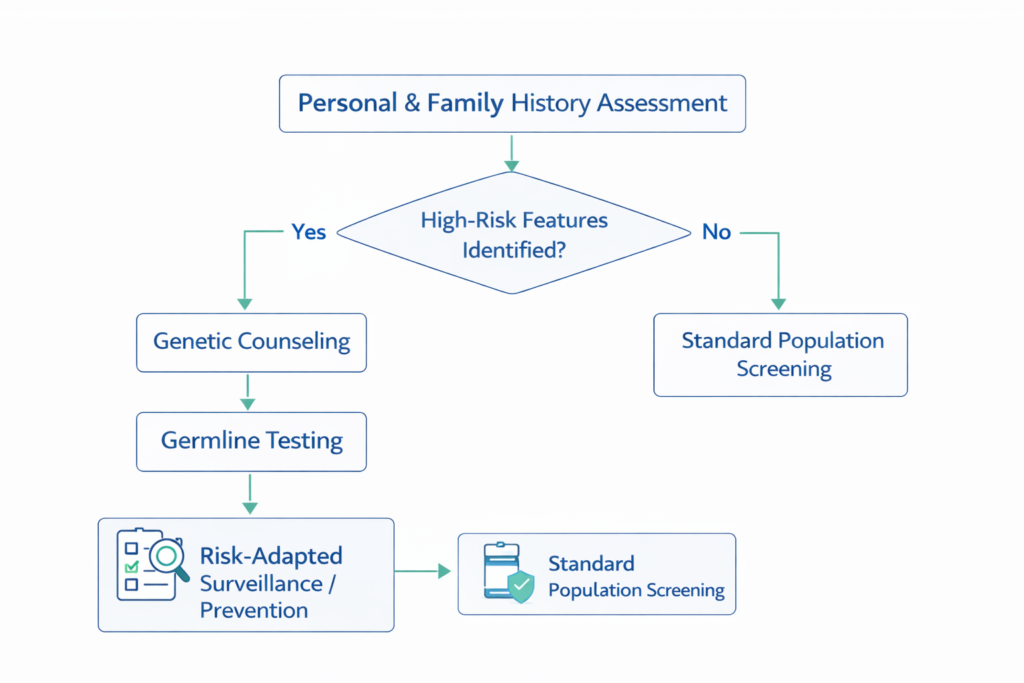
Step 1: Assess detailed three-generation family history.
Step 2: Identify high-risk patterns or early-onset disease.
Step 3: Refer for genetic counseling.
Step 4: Perform germline testing if criteria met.
Step 5: Implement surveillance or preventive strategy based on results.
How Does Genetic Testing Influence Treatment?
Beyond risk prediction, germline mutations may influence therapy selection.
- PARP inhibitors in BRCA-mutated cancers
- Immunotherapy in Lynch-associated tumors
- Risk-reducing surgeries in high-risk carriers
Testing therefore bridges prevention and precision treatment.
When Is a Second Opinion Valuable?
Variant interpretation can be complex. Variants of uncertain significance require careful clinical correlation.
- Before prophylactic surgery decisions
- When results are inconclusive
- In rare hereditary syndromes
Closing Perspective
Genetic testing in oncology represents a shift toward anticipatory medicine. By identifying inherited risk early, clinicians can implement evidence-based surveillance and prevention strategies. Structured evaluation ensures testing is precise, ethical, and clinically meaningful.
Frequently Asked Questions
Is genetic testing recommended for everyone?
No. Testing is guided by personal and family risk patterns.
Does a positive result mean cancer is certain?
No. It indicates elevated risk, not inevitability.
Does a positive result mean cancer is certain?
It cannot prevent mutations but allows preventive strategies and surveillance.
What is the difference between germline and somatic testing?
Genetic testing for cancer risk involves assessment of 2 types of testing. Germline testing identifies inherited risk; somatic testing evaluates tumor-specific mutations.
Should unaffected family members get tested?
If a pathogenic mutation is identified in the family, targeted testing may be appropriate. There is another variant – VUS for which nothing has to be done.
At what age should someone consider genetic testing for cancer risk?
There is no universal age for testing. In families with known hereditary mutations, testing is often considered in early adulthood, particularly when results would influence surveillance or preventive decisions. Testing in minors is generally reserved for syndromes where childhood cancer risk is significant.
Related Articles
• Understanding BRCA Mutations in Cancer Risk
• Molecular Profiling in Modern Oncology
• Structured Second Opinions in Cancer Care
Pingback: individualizing-cancer-treatment-in-the-elderly-and-young-adults