Biomarker-driven therapy is a precision oncology approach where cancer treatment is selected based on specific molecular characteristics of a tumor rather than its location alone. These biomarkers may include gene mutations, protein expression levels, or genomic signatures. By identifying actionable targets, therapy becomes more individualized, potentially improving outcomes and minimizing unnecessary toxicity. Additionally, research continues to demonstrate the efficacy of this approach across various cancer types, emphasizing the importance of continuous advancements in molecular diagnostics and treatment modalities.
Modern oncology increasingly integrates molecular diagnostics into treatment planning. This strategy represents a shift from organ-based treatment models to biology-driven decision-making. With the advent of new technologies, oncologists can now analyze tumors at a molecular level, allowing for tailored therapies that align closely with the unique characteristics of each patient’s cancer. This evolution in treatment planning is not only revolutionizing patient outcomes but also enhancing the overall understanding of cancer biology.
As a consultant medical oncologist practicing precision oncology, I routinely incorporate biomarker analysis into structured treatment planning across tumor types.
In addition to improving individual treatment plans, biomarker-driven therapy also paves the way for clinical trials that specifically target those biological markers, allowing for a more profound understanding of treatment efficacy and patient response. For example, studies have shown that patients with specific mutations may respond exceptionally well to certain targeted therapies, highlighting the need for comprehensive biomarker testing as part of standard care.
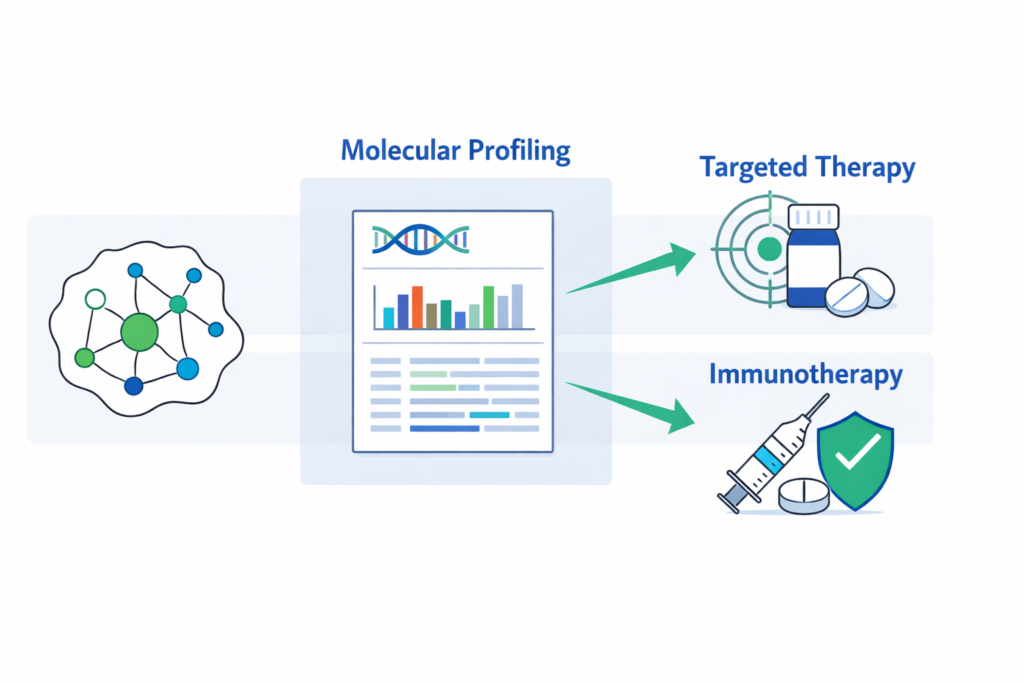
What Exactly Is a Biomarker in Cancer Care?
A biomarker is a measurable biological characteristic that provides information about cancer behavior or treatment response. Various types of biomarkers exist, each playing a critical role in the oncological landscape. For instance, genetic mutations can indicate the likelihood of developing certain cancers, while protein markers can provide insights into tumor aggressiveness and treatment sensitivity. As research progresses, the utility of biomarkers continues to expand, underscoring their significance in personalized medicine.
- Genetic mutations (e.g., actionable driver mutations)
- Protein overexpression (e.g., receptor amplification)
- Immune markers (e.g., PD-L1 expression)
- Genomic instability signatures
Not all biomarkers are actionable. The clinical relevance depends on validated evidence linking the marker to therapeutic benefit. Continuous research and validation are necessary to ensure that these biomarkers accurately reflect the biology of different cancer types, providing oncologists with reliable data to inform treatment plans.
How Does Biomarker-Driven Therapy Change Treatment Selection?
Traditional oncology often relied on tumor site and stage alone. Biomarker-driven therapy introduces biological stratification. This new approach allows for a more nuanced understanding of tumor behavior, enabling oncologists to choose therapies based on individual patient profiles rather than solely on physical tumor characteristics. For patients, this means treatments that are more effective and potentially less toxic.
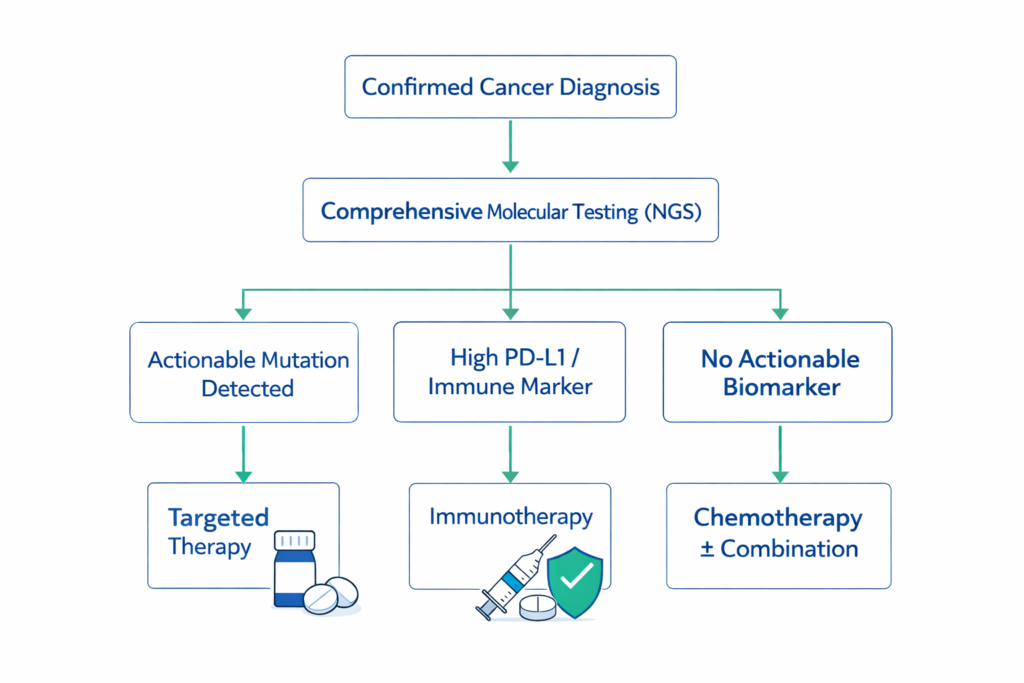
- If actionable mutation detected → Then targeted therapy preferred.
- If high immune expression markers → Then immunotherapy considered.
- If no actionable markers → Then chemotherapy ± combination strategies.
- If resistance mutation identified → Then next-line targeted sequencing.
This structured logic improves therapeutic precision and sequencing, marking a significant advancement in personalized cancer treatment.
Which Cancers Commonly Use Biomarker-Guided Strategies?
Biomarker-driven therapy is now standard of care in multiple malignancies, enhancing treatment outcomes across various cancer types:
- Non-small cell lung cancer
- Breast cancer (HER2, hormone receptors)
- Colorectal cancer (RAS, BRAF)
- Melanoma (BRAF mutations)
- Certain hematologic malignancies
The scope continues to expand as next-generation sequencing panels become more accessible, encouraging further integration of biomarker-driven therapies into routine practice.
Structured Decision Tree in Biomarker-Driven Oncology
This structured decision-making framework allows for a systematic approach to treatment planning, ensuring that each patient’s unique tumor profile is considered when developing a treatment plan. Understanding the interconnections between various biomarkers and potential therapies can lead to more effective and personalized care.
When Is a Second Opinion Valuable in Biomarker-Driven Therapy?
Interpretation of genomic reports can be complex. Variant classification, resistance mutations, and off-label targeting require expertise. It’s essential for patients undergoing biomarker testing to understand their results fully and explore all options available to them. Seeking a second opinion can provide reassurance and potentially reveal alternative treatment strategies that may be overlooked.
- Before starting expensive targeted therapy
- When genomic variants are labeled “uncertain significance”
- When treatment sequencing options differ
- In rare or advanced-stage cancers
A structured second review may significantly influence long-term outcomes, emphasizing the importance of comprehensive evaluation and informed decision-making in cancer treatment.
Frequently Asked Questions
Is biomarker testing required for every cancer patient?
Not universally. It depends on cancer type, stage, and evidence-based guidelines. As the field advances, the criteria for testing are evolving, highlighting the need for oncologists to stay updated on best practices.
Does biomarker-driven therapy replace chemotherapy?
No. It complements existing modalities and may reduce chemotherapy use in selected cases. The balance between targeted therapies and traditional chemotherapy is a key area of ongoing research, as treatment strategies continue to evolve.
How long does molecular testing take?
Turnaround time varies but typically ranges from 7–21 days depending on platform. However, advancements in technology are decreasing these times, allowing for quicker access to crucial treatment information.
Are targeted therapies less toxic?
They may have different toxicity profiles but are not universally safer. The side effects can vary significantly based on the type of targeted therapy used, necessitating careful monitoring and adjustment of treatment as necessary.
Can resistance develop to targeted therapy?
Yes. Secondary mutations may emerge, requiring re-biopsy or treatment modification. Understanding resistance mechanisms is a vital component of ongoing cancer research, shaping the future of targeted therapies and patient management.
Related Articles
In summary, the integration of Biomarker-Driven Therapy into cancer care is not merely a trend but a fundamental transformation that enhances the way oncologists approach treatment. With continuous advancements in research and technology, the potential for personalized cancer treatment will only grow, making it essential for patients to engage in discussions about biomarker testing with their healthcare providers.
When Should Patients Undergo Genetic Testing for Cancer Risk?
When and Why Genetic Testing Matters in Cancer Treatment and Prevention
What Is Biomarker-Driven Therapy in Modern Oncology?
Closing Perspective
Biomarker-driven therapy represents the evolution of oncology toward biological precision. As molecular understanding deepens, treatment decisions increasingly rely on genomic intelligence rather than anatomical classification alone. The future of cancer care is structured, evidence-driven, and individualized. A commitment to research and education will further enhance the effectiveness of biomarker-driven therapies, ultimately leading to improved patient outcomes and survival rates.
Educational Disclaimer: This article is for educational purposes only and does not substitute individualized medical consultation or treatment planning. Always consult with a healthcare professional regarding medical advice or treatment decisions.
Pingback: Molecular Subtypes of Breast Cancer
Pingback: Immunotherapy in Low Pd-L1 expression
Pingback: Molecular Subtypes in Cancer:Biology Matters More Than Stage
Pingback: Targeted Therapy and Chemotherapy : Understanding the Difference